Can a Camera, a QR Code and Some Bubbles Test For E. Coli In Our Food?
MIT researchers are pursuing a newer, faster test for foodborne pathogens
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/7c/ba/7cbabb2e-9eb4-4f82-af93-53d0a2f961f0/mit-foodborn-1.jpg)
Here’s how it works: Mix some tiny, special-made bubbles in with liquid you think might contain E. coli bacteria. Place a QR code beneath the petri dish, and turn on your phone’s camera. If the phone can read the code, it’s safe. If not, there’s E. coli.
This is the finding of research published in the American Chemical Society’s new journal, Central Science. The technique hinges on the design of the microscopic droplets, and its authors say it has the potential to vastly reduce the amount of time required to test food.
Food poisoning is a big problem, even in the U.S., with E. coli causing 73,000 illnesses and 60 deaths per year, according to Centers for Disease Control and Prevention data from 1999. It’s basic, but speeding up testing should mean more testing gets done.
“The big problem is, when you’re manufacturing food, if you don’t have something that is basically at the time scale of your manufacturing process, you’re having to hold product up in a storage facility [for testing],” says Tim Swager, a chemistry professor at MIT and author of the study. “You need something that’s virtually minutes, or maybe a couple hours, not a day, or tens of hours. And that’s where the current state of the technology is right now. This is too slow, and very costly.”
Swager’s team blends two types of material into micron-scale droplets called Janus emulsions. They begin with two materials, hydrocarbon (H2O bonded with carbon, like you get in fuel gases) and fluorocarbon (fluorine bonded with carbon, a material used in fishing line). They heat the two fluids up, and force them together through tiny channels, injecting them into flowing water sort of like a pipe dumping in a river. As the particles cool, they form spheres that are half hydrocarbon, half fluorocarbon.
To these droplets, the scientists attach a plant protein called lectin, which binds to E. coli. Normally, the heavier fluorocarbon side of the droplets keeps them level, all with their hydrocarbon hemispheres facing upward. In that state, they act like a lens with an infinite focal length; light travels through in a straight line. But when the lectin binds, the bacteria that attaches changes the balance of the droplets, causing them to tip on their side. When that happens, the refraction scatters light, blocking out whatever’s underneath.
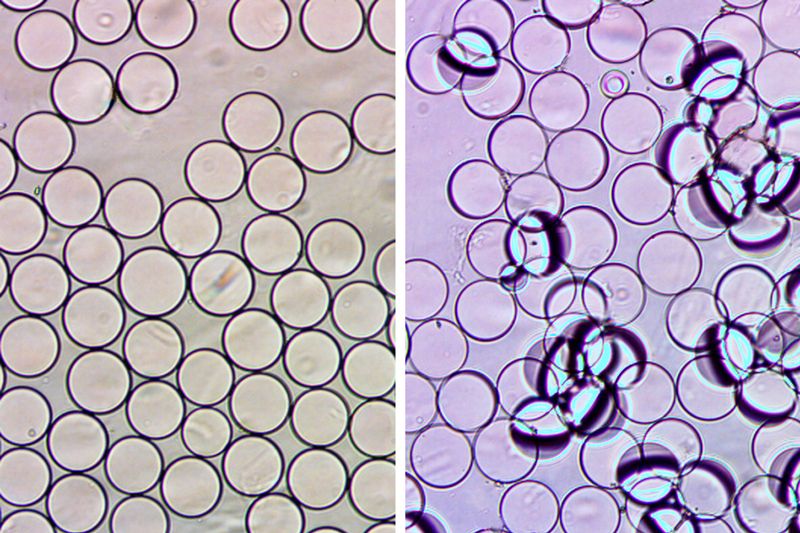
The researchers have tested this technique on several types of benign E. coli, and plan to expand the technique to other bacteria, or even other types of pathogens.
“The fact they can respond so well, they can tilt, and we can reorient them and they behave like lenses, and that we’re using gravity to align them, these are a lot of really unusual ingredients, but it makes for a really powerful platform,” says Swager.
Applying the technology to pathogenic strains is feasible, but you’d need a different binding structure for each one, says John Mark Carter, a former food borne contaminants researcher at the USDA who now consults in the same industry.
“It’s really not as easy as it sounds,” Carter says. “Food contains a lot of things that bind a variety of surfaces nonspecifically.”
He adds that the droplets must be balanced precisely, which the researchers were able to do, but it becomes much more problematic in real-life food-safety testing. Carter is surprised that the researchers are proposing a food test at this point. “You really shouldn’t talk about food until you do experiments with food,” he says.
Additionally, sensitivity limits for E. coli in food are much lower than this technique can yet offer. Swager was able to detect the presence of E. coli when there are around 10,000 cells per mL of solution. In 2010, the FDA reduced the amount of non-toxic E. coli it allowed in cheese (a sort of general sanitation limit) from 100 MPN (most probable number) per gram to 10 MPN. Last year, the agency backed off on that, saying it hadn’t had an impact on public health, but for food safety and toxic E. coli, tolerance is zero. There’s no tech that can detect a single E. coli cell, which is why the present standards rely on growing colonies in a dish.
“One bacteria is enough to kill you,” says Carter. “If it’s a very high concentration of bacteria you can detect it without amplification. But almost everybody grows it…you have to grow it, because you can’t detect one bacteria.”
If Swager’s tech is to become widespread in the industry, these issues must be addressed, and then side-by-side studies run to compare it with the current standards. Absent that, there could be applications for internal quality control among the food industry (though detection sensitivity would still be an issue).
“Eggo waffles had a recall [in 2016]. It wasn’t a huge recall, but it was listeria,” says Swager. “When my kids were little, I used to give them Eggo waffles, and they’d go out the door on their way to school. But you know, you have a product recall like that, how long is it going to be before parents are going to feed their kids Eggo waffles? So the implications to the brands are also really high.”