Why Immunity to the Novel Coronavirus Is So Complicated
Some immune responses may be enough to make a person impervious to reinfection, but scientists don’t yet know how the human body reacts to this new virus
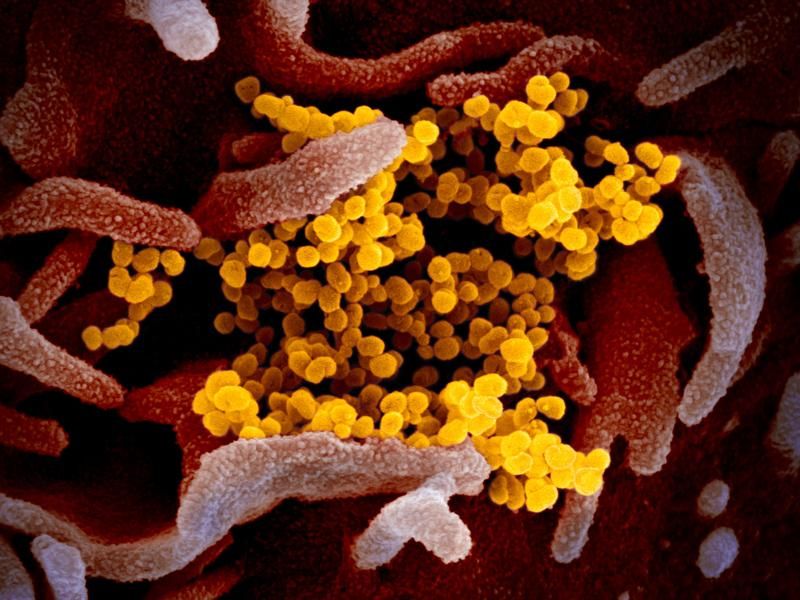
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/26/aa/26aa2fcb-6815-4efe-961d-616e5fe39a99/sars-cov-2_scanning_electron_microscope_image.jpg)
Even before the blood left his arm, André Valleteau suspected he knew what his doctors would find.
Just weeks before, the 27-year-old from Toronto had tested positive for SARS-CoV-2, the coronavirus that causes COVID-19. The symptoms hit him hard: headache, cough, sore throat and fatigue that relegated him to his bed 15 hours a day. “It didn’t matter how many times I napped,” he says. “I was tired until the next time I napped again.”
Valleteau, a researcher coordinator at a pharmaceutical company, spent two weeks self-isolating and recovering, then decided he wanted to help others do the same. He contacted a local researcher and offered up his blood—along with the disease-fighting antibodies that likely teemed within. Indeed, Valleteau’s blood tested positive for antibodies against SARS-CoV-2, and a team of scientists is now studying molecules from patients like Valleteau in the hopes they can inform the development of drugs or vaccines to vanquish the virus.
Antibodies, which the body makes in response to dangerous microbes like SARS-CoV-2, are crucial for defending against disease. Many can glom onto pathogens and subdue them before they have a chance to encounter vulnerable human cells. Antibodies are also evidence: Some COVID-19 tests target these molecules because they show that someone has previously been infected with SARS-CoV-2. (And as previously reported, the possibility of false negatives or false positives, which are more common with some tests than others, can sometimes muddle attempts to pinpoint past infections.)
Even then, while a positive antibody test (also called a serology test) can say a lot about the past, it may not indicate much about a person’s future. Researchers still don’t know if antibodies that recognize SARS-CoV-2 prevent people from catching the virus a second time—or, if they do, how long that protection might last.
Immunity isn’t binary, but a continuum—and having an immune response, like those that can be measured by antibody tests, doesn’t make a person impervious to disease. “There’s this impression that ‘immunity’ means you’re 100 percent protected, that you’ll never be infected again,” says Rachel Graham, an virologist studying coronaviruses at the University of North Carolina’s Gillings School of Global Public Health. “But having immunity just means your immune system is responding to something”—not how well it’s poised to guard you from subsequent harm.
It takes a symphony of cells
In discussions of immunity, antibodies often end up hogging the spotlight—but they’re not the only weapons the body wields against invaders. The sheer multitude of molecules at work helps explain why “immunity” is such a slippery concept.
When a pathogen infiltrates the body, the immune system mounts a defense in two acts. First comes the innate immune response, a blunt, broad-acting ensemble that attacks any invader that doesn’t resemble a normal-looking human cell. Slower but more specific is the adaptive immune response, a second wave of assailants the body custom-builds to recognize unique features of the infectious microbe.
This second wave includes antibodies, which are manufactured by immune cells called B cells. Some antibodies are potent weapons that curb a microbe’s capacity to latch onto and enter cells, while others simply flag germs or infected cells for destruction by other parts of the immune system. The former category, called neutralizing antibodies, are necessary to combat most of the pathogens that plague humans, and their production is generally considered a hallmark of a good vaccine, says Sallie Permar, a virologist and vaccine expert at Duke University says.
Although antibodies have a short lifespan, disappearing from the blood after a few weeks or months, the immune system retains some of the B cells that produce them. If the same germ returns, these cellular factories will whip up a big batch of antibodies to wage a second war. But antibodies alone aren’t enough to quash an infection, says Diane Griffin, an immunologist at Johns Hopkins University’s Bloomberg School of Public Health. “You need an orchestra of responses [for protection] to really be effective.”
T cells—another subset of the adaptive response—are often left out of conversations about immunity.
“You can’t have a great antibody response without T cells,” says Akiko Iwasaki, a virologist and immunologist at Yale University. Among a slew of helpful functions, T cells help young B cells mature into antibody-making machines. “These things really go hand in hand.”
T cells are also formidable fighters in their own right. In a bid to stop the spread of a pathogen throughout the body, some T cells will trigger infected cells to self-destruct. Others linger after an illness has resolved, patrolling tissues so germs can’t reestablish a foothold. (One of the reasons that HIV is such a devastating diagnosis is that the virus destroys some of the body’s T cells.)
Studies of other coronaviruses, including the ones that cause SARS and MERS, show that T cells play integral roles in stamping out sickness, says Stephanie Langel, a virologist and immunologist at Duke University. It’s likely that the same will hold true for SARS-CoV-2. Compared to antibodies, however, T cells—which often hole up in hard-to-reach tissues like the lungs—are more difficult to extract and analyze. That makes T-cell detection unlikely to play much of a role in clinical tests for immune responses against SARS-CoV-2. For researchers, though, T cells “represent a wealth of knowledge” about how our immune systems deal with the new coronavirus, Langel says.
Antibodies aren’t perfect
Even the most sensitive laboratory tests have their limits, and finding antibodies against SARS-CoV-2 is no guarantee that those molecules are high-quality defenders or that a person is protected from reinfection.
Most commercially available antibody tests search a person’s blood for antibodies that can recognize SARS-CoV-2’s spike protein, the molecule the virus uses to attach to human cells. Some (but not all) of these assessments can tell how many antibodies a person is producing—the more the better, generally speaking. But typical tests don’t determine whether the antibodies are neutralizing. To come to that conclusion, researchers must mix antibodies with viruses and check whether they stop the pathogens from invading human cells in a laboratory under strict safety protocols.
Though non-neutralizing antibodies can play less direct roles in incapacitating a pathogen, many are duds that have no effect on microbes. Some may even chauffeur active viruses into healthy cells, inadvertently accelerating infection. “Essentially, the antibody helps the virus replicate,” Permar says. This rare phenomenon, called antibody-dependent enhancement, has been observed with the viruses that cause dengue and Zika.
Why antibodies can be so inconsistent is still a mystery, in part because laboratory experiments can’t recreate the conditions these molecules experience in the body, says Marcia Goldberg, a microbiologist at Harvard University. “It’s really important to actually test how well antibodies are working in people.” (One way to do so involves administering antibodies to patients battling COVID-19, something that’s being trialed in hospitals around the world.)
Even antibodies with known neutralizing powers aren’t foolproof. Donna Farber, an immunologist at Columbia University who studies T-cell responses against airway viruses, says that some patients with high levels of neutralizing antibodies in their blood still succumb to COVID-19, another hint that other parts of the immune system are needed to reliably defeat this disease.
That’s why antibody tests shouldn’t be overinterpreted, Iwasaki says. One common misconception is that a positive antibody test means a person no longer has the virus in their system, which isn’t necessarily the case. Antibodies are often roused about a week into a new infection, potentially overlapping with a pathogen’s tenure in the body. Diagnostic tests that search for the virus’ genetic material can help tease that timeline apart, but even these assessments can yield incorrect results.
“So much nuance is being lost by just saying ‘serology [antibody] positive’ and ‘serology [antibody] negative,’” Iwasaki says.
A range of responses
A person who has recovered from their first brush with a new pathogen like SARS-CoV-2 may travel one of several immunological routes, Goldberg says—not all of which end in complete protection from another infection.
One possibility is that the immune system does a great job of cataloguing the invader’s unique features. That intel will get stored in an army of B and T cells that will rally to fight the second time a germ comes around. B cells in particular play a starring role in this scenario, pumping out neutralizing antibodies that can sequester and disable a pathogen before it even has the chance to enter a host cell, Iwasaki says. This phenomenon, called sterilizing immunity, make people essentially resistant to reinfection. The body may rouse this reaction in response to microbes like the virus that causes measles, which tends to be a one-and-done infection for most people.
But not all pathogens prompt such a robust response—and not everyone reacts the same way to a given microbe. People can experience varying shades of partial protection in the wake of an infection, Goldberg says. In some cases, a bug might infect a person a second time but struggle to replicate in the body, causing only mild symptoms (or none at all) before it’s purged once more. The person may never notice the germ’s return. Still, even a temporary rendezvous between human and microbe can create a conduit for transmission, allowing the pathogen to hop into another susceptible individual.
Under rarer circumstances, patients may experience symptoms that are similar to, or perhaps even more severe, than the first time their body encountered the pathogen.
That doesn’t mean people are doomed to experience the same diseases over and over. “The word ‘immune’ makes it sound like the virus gets close to your body, hits a wall, and has to turn away and go find someone else,” says Allison Roder, a virologist at New York University. But even partial protection from the immune system will curtail the amount of pathogen in a person’s body, and, by extension, the likelihood of transmission.
None of these protective states are necessarily permanent or mutually exclusive. A person’s immunity to a pathogen can wane over the course of months or years, eventually dropping below a threshold that leaves them susceptible to disease once again. Researchers don’t yet know whether that will be the case for SARS-CoV-2. The widespread deployment of accurate antibody tests, which can track both where the virus has been and how people are faring after having it, may help answer that question. Scientists are also trying to determine the antibody levels that correlate with protection against reinfection and how durable those responses are over time.
Finding those answers will be a boon for vaccine development, Permar says. “The dream of every vaccine is to be able to say, ‘We need this level of antibody.’ Then vaccines can chase that endpoint. Until we know that benchmark… we’re operating in the dark.”
So far, early studies in both humans and animals suggest exposure to SARS-CoV-2 marshals a strong immune response. But until researchers have more clarity, Graham advises continued vigilance—even for those who have gotten positive results from antibody tests, or have other reason to believe they were infected with COVID-19.
Valleteau, who has received clinical confirmation the coronavirus is no longer in his system, is still practicing physical distancing, frequently washing his hands and wearing protective gear like face coverings. He’s also taking extra precautions around the patients he works with, many of whom have diabetes, a condition that can increase the risk of complications from COVID-19.
“This is not a free pass to act like nothing is going on,” he says. “Just because you’ve had it and recovered, you’re not absolved of social responsibility.”
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/accounts/headshot/10172852_10152012979290896_320129237_n.jpg)
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/accounts/headshot/10172852_10152012979290896_320129237_n.jpg)