Human Cells Ward Off Bacterial Invaders With a Protein That Behaves Like Soap
Researchers discover that immune cells aren’t the body’s only line of defense against bacterial pathogens
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/51/f0/51f07ee4-e586-4dfb-b65b-ca19af0b0694/3d_slice_2_715_px.jpg)
When bacteria get past our immune cells, the body has one more trick up its sleeve. The nonimmune cells join the fight by unleashing a soap-like protein that takes a bite out of the walls of the bacterial invaders.
“We were a bit surprised to find detergent-like activity inside human cells,” John MacMicking, an Yale University immunologist, tells Jonathan Lambert of Science News.
The new study, recently published in the journal Science, provides a glimpse of the complex strategies that the human body employs to defend itself against pathogenic infiltrators. While immune cells and antibodies hog most of the attention from the research community, the new research turns the spotlight onto the oft-overlooked cellular defenses of nonimmune cells. These cells are important players of “an ancient and primordial defense system,” MacMicking tells Science News.
The bacteria-slaying, soap-like protein is an apolipoprotein called APOL3. Apolipoproteins are normally used for ferrying lipids molecules around the body to be used as energy or for building cells, reports Abby Olena for The Scientist. Like soap, the APOL3 molecules contains a water-loving end and a fat-loving end, so it can bind to the lipid membranes of bacteria and dissolve chunks of the wall into the intracellular fluid. Moreover, MacMicking’s team found APOL3 in a variety of tissues throughout the body, so the researchers suspect it could offer wide protection.
To study this cellular defense mechanism, MacMicking and his colleagues infected human epithelial cells with Salmonella, the germ responsible for food-poisoning. The bacterium possesses two membranes: an outer one for armor and an inner one as protection against threats such as antibiotics, according to a Howard Hughes Medical Institute statement.
After screening over 19,000 human genes, the researchers found that APOL3 works in concert with the GBP1 protein to wreck Salmonella. With the aid of a high-resolution microscope, the researchers pieced together how the proteinaceous pair defend against bacteria. Upon receiving red-alert signals from the immune system, non-immune cells churn out both proteins en masse. GBP1 lands the first blow on Salmonella’s outer membrane, allowing APOL3 to pass through and break apart the inner membrane. The killer APOL3 protein then swarms the bacteria and destroys them.
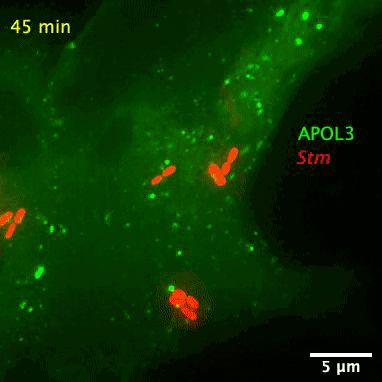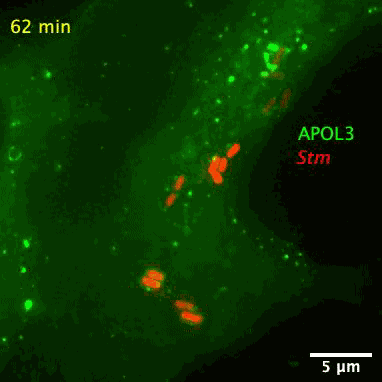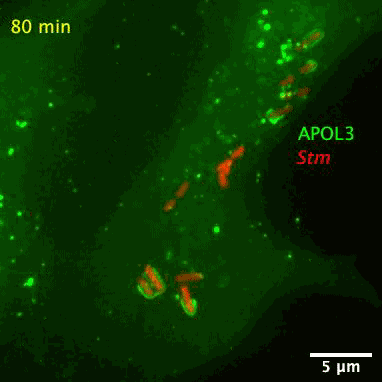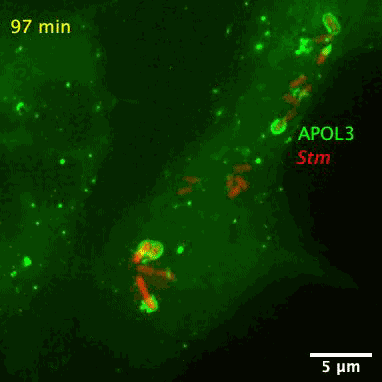
The researchers also found APOL3 targets the lipids in bacterial membranes and spares its own host. The wily protein distinguishes the two kinds by avoiding cholesterol, which is often found in the walls of mammalian cells.
“Everything about these findings is supercool,” evolutionary immunologist Jessica Brinkworth of the University of Illinois at Urbana-Champaign who wasn’t involved in the study, tells Science News. She calls APOL3’s selectivity of its bacterial targets “a beautiful thing.”
MacMicking says his next move is to uncover the potential defense strategies of other apolipoproteins related to APOL3. He also hopes the study’s findings can guide future efforts to develop new treatments for bacterial infections, per the statement.
“This is a case where humans make their own antibiotic in the form a protein that acts like a detergent,” says MacMicking in the statement. “We can learn from that.”
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/accounts/headshot/kim.png)
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/accounts/headshot/kim.png)