Breathtaking Bubbles, Butterfly Wings, and a Glowing Atom Take Top Prizes in Science Photo Contest
The images celebrate the depth and beauty of the physical sciences
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/4a/75/4a75c99f-93b1-4397-97b9-3daae3d1eb51/a7639384-2b5f-4294-b3529347d576cd4e_featuretwocolumnwide.jpg)
Earlier this week, the U.K.-based Engineering and Physical Sciences Research Council (EPSRC) revealed the winners of its national science photography competition. Selected from 100 entries, all of which received EPSRC funding, the photos demonstrate the breadth and beauty found in physical science research—healthcare, materials science, mathematics and chemistry.
"Not only do we have really strong, attractive photographs, the stories behind them about the research and why it is being done are inspiring." Dame Ann Dowling, president of the Royal Academy of Engineering and one of the judges says in a press release. "Much of this work will lead to innovations that transform lives and, in this Year of Engineering, it’s marvelous to see these great examples of transformational research.”
Single Atom in Ion Trap - First place equipment and facilities and overall competition winner
It's fairly common knowledge that atoms as so tiny they can’t be seen by the naked eye. Even seeing them with a sophisticated microscope is quite a feat. But David Nadlinger from the University of Oxford figured out a way to make visible what is usually too small to be seen. He set up an ion trap inside a vacuum chamber in his lab, then hit a strontium atom with a blue violet laser. The atom then re-emitted enough of the light that a long exposure camera shot could show a single atom.
“The idea of being able to see a single atom with the naked eye had struck me as a wonderfully direct and visceral bridge between the miniscule quantum world and our macroscopic reality,” Nadlinger says in the press release. “A back-of-the-envelope calculation showed the numbers to be on my side, and when I set off to the lab with camera and tripods one quiet Sunday afternoon, I was rewarded with this particular picture of a small, pale blue dot.”
That pale blue dot, just a pixel or two on a computer screen, is a little hard to make out. But it’s worth the squinting to “see” an atom. “It’s exciting to find a picture that resonates with other people that shows what I spend my days and nights working on,” Nadlinger tells Ryan F. Mandelbaum at Gizmodo.

In a kitchen far far away... - First place eureka and discovery
Soap bubbles are a little freaky if you look closely. The rainbow colored surfaces swirl and dance before popping. Li Shen and his colleagues at the Imperial College London took a closer look at just how the tiny bubbles work, using a rig Shen made of household items. “The [photo] was taken in my kitchen using a simple bubble-film apparatus I made from a funnel and some dishwashing liquid, using the technique of interferometry, whereby you use colors to distinguish between the thickness of the bubble membrane on the film,” he says in a press release. The rig also used cookie tins, a water bottle and an oven tray.
Shen didn’t get these items back to cook with for a while—the setup, photography and videography of the bubbles took about a month. While the shoot was made from simple objects, the bubbles are anything but. Shen and his team found a very complex set of fluid dynamics that govern how the soap bubbles form, evolve and eventually pop.

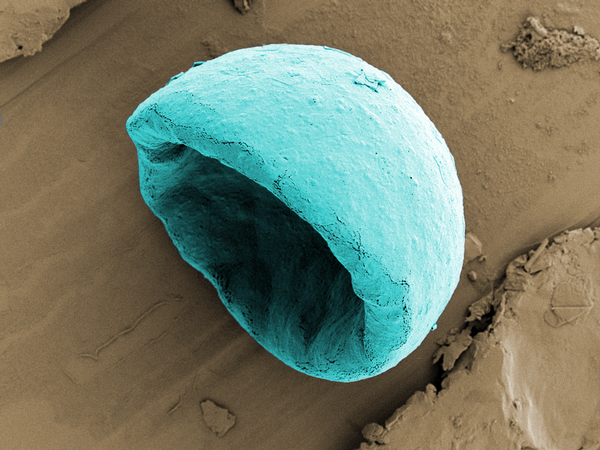
Microbubble for drug delivery - First place innovation
One of the problems with all the wonder drugs science comes up with is getting them where the need to go. In many cases, powerful drugs are absorbed by the entire body, sometimes causing terrible side effects or damage instead of heading straight to the target organ, tumor or infection. That’s why researchers have been working on a concept called microbubbles in recent years. According to The Yorkshire Evening Post, the bubbles contain the medicine—like a chemotherapy drug—in a shell. When the bubbles are injected into the blood stream, they don't release the drug right away. Instead, a technician monitors them, waiting for them to collect at a tumor site before “popping” them using ultrasound.
Estelle Beguin of the University of Oxford imaged one of the microbubbles, just a few microns across, using a Transmission Electron Microscope. This particular bubble has a gas core and is coated with liposomes, or small spherical sacs, containing a drug.

Natures Nanosized Net for Capturing Color - First place weird and wonderful
Butterflies, of course, are known for their beautiful array of colors. But the dazzling hues are not all made the same way. Anyone who has picked up a dead monarch knows that the orange and red colors are carried by a pigment that readily rubs off on your fingers. Liz Langley at National Geographic explains that other colors, including blue, purple and white are structural, created by the scattering of light by features on the insect’s wings. Bernice Akpinar at Imperial College London used atomic force microscopy to get an up close view of those micro-meter scale structures. Her winning image shows the 1 micron ridges connected by cross ribs on a butterfly wing which produce a brilliant iridescent color that never fades. Research into structural color, which is also found on some bird feathers and other insects like peacock spiders, could lead to paints or coatings that don't use pigments and never lose their shine.
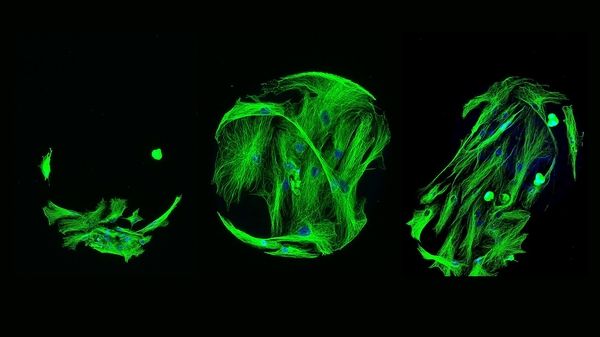
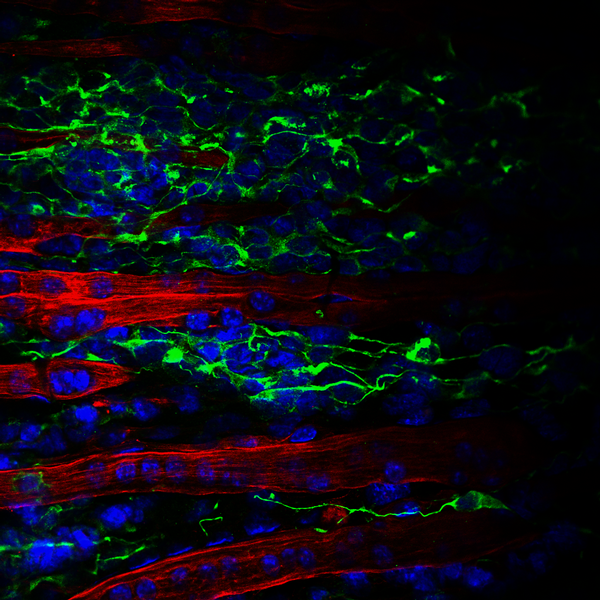
Check out a few more of the winners below: