VIDEO: Functional Liver Tissue Can Now Be Grown From Stem Cells
By mixing different types of stem cells in petri dishes, researchers created liver “buds” that effectively filtered blood when implanted in mice
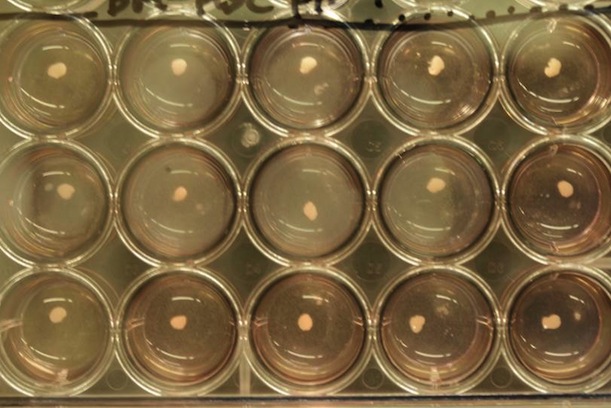
Liver “buds”—small lumps of functional three-dimensional human liver tissue—were grown from a mix of stem cells. Photo by Takanori Takebe
If you’re suffering from liver failure and are in need of a transplant, the odds are bleak. Acute liver failure can cause death within a few months or weeks from the time symptoms first appear, and chances of getting a liver transplant are slim—there are nearly 17,000 people in the U.S. waiting for a liver right now and the average wait time can approach a year in length.
This is the reason that scientists are striving to figure out alternate means of generating replacement livers, mostly by manipulating induced stem cells to develop liver tissue. Recently, a team of scientists from Japan took a huge step forward towards this goal, using stem cells to grow small, rudimentary lumps of liver that functioned effectively when implanted in mice, producing proteins and filtering out chemicals as normal livers do.
The group presented preliminary reports of their success last year at the annual meeting of the International Society for Stem Cell Research, but didn’t reveal the full details until today, in a study published in Nature. Although it’s far from certain that their method will ever result in a replacement liver—and even if it does, it’ll be years before it’s proven safe and effective for humans—the accomplishment represents the first-ever functional human organ, albeit small-scale, that was grown solely from stem cells.
The researchers, led by Takanori Takebe of Yokohama City University, worked with induced pluripotent stem cells, which are like embryonic stem cells in that they’re capable of developing into any possible type of human tissue, but are produced by manipulating normal adult somatic cells. Due to the ethical concerns associated with harvesting stem cells from human embryos—along with the fact that, in the U.S., there are numerous legal restrictions against doing so—most scientists in the field are now using these sorts of stem cells instead.
The scientists started by using particular chemical signals to prompt these stem cells, growing in a petri dish, to develop into generalized liver cells known as hepatocytes, as has been done in their prior research. Their real trick, though, was what came next. To mimic the development of liver tissue in normal human embryos, they mixed in two other types of cells: endothelial cells (which line the inside of blood vessels) and mesenchymal stem cells (which can develop into fat, cartilage or bone).
All these cells were placed in a two-dimensional arrangement in the petri dishes, but over the next two days, they self-organized into three-dimensional liver “buds,” which were roughly five millimeters long and resemble the early phase of liver development in healthy human embryos, roughly five weeks after fertilization. Under a microscope, the buds were shown to have the same organizational mix of hepatocytes and blood vessels normally present in liver tissue.
The team reportedly tried out hundreds of various combinations of cell types added at different stages and in different quantities before arriving at the precise cocktail that led to the growth of liver buds, a process that took more than a year. They released a time lapse video that shows the successful cell mixture spontaneously absorbing nutrients and growing into a liver bud (a specially-stained version is shown on the left):
They put these liver buds to the test by implanting them in mice, either inside their abdominal cavities or in their skulls. A variety of tests showed that these rudimentary livers were able to perform all the same functions as mature human livers—producing the same proteins, filtering out all the same substances and, crucially, metabolizing particular chemicals that human liver tissue can normally deal with but mouse liver tissue cannot.
There’s still a tremendous amount of research necessary before this sort of process could be used to produce replacement liver tissue for humans—Takebe guesses that it’ll be roughly ten years before the first human clinical trials are able to begin. For the researchers, the next steps are generating liver buds that resemble normal liver tissue even more closely (complete with bile ducts and other structures) and producing greater numbers of buds, as they estimate that somewhere on the order of tens of thousands would be necessary to grow one liver, the largest internal organ of the human body.
Perhaps the most exciting aspect of this research, though, is its potential universality: The researchers speculate that this sort of method could someday be used to grow all sorts of organ tissue, including that of the pancreas and kidneys. In the short term, the technique could also have innovative applications—for example, liver buds alone could be used to test the toxicity of drugs in development, because the full organ’s internal structures aren’t necessary to determine which chemicals can’t be broken down in the body.
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/accounts/headshot/joseph-stromberg-240.jpg)
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/accounts/headshot/joseph-stromberg-240.jpg)