The Latest Cure for Acne: A Virus
Researchers are looking into a naturally-occuring virus which preys upon the skin bacteria that trigger outbreaks of acne
![]()
A new anti-acne approach acts upon Propionibacterium acnes, the naturally-occuring bacteria that cause outbreaks. Image via Wikimedia Commons/Bobby Strong
Acne afflicts nearly 90 percent of Americans at some point in their lives, but scientists have made surprisingly little headway in understanding and combating the skin condition. For sufferers of severe acne, the treatments available— benzoyl peroxide, antibiotics and Accutane—are limited in effectiveness and can cause a range of undesirable side effects.
New research, though, is pointing towards a novel approach that could someday serve as a solution: the use of viruses to attack the bacteria on the skin that cause acne breakouts. After studying the Propionibacterium acnes phages virus and sequencing its DNA, a team of researchers from the University of California, Los Angeles and the University of Pittsburgh believes that it could be an ideal candidate for the development of a new sort of anti-acne therapy. Their findings were published today in the journal MBio.
“Acne affects millions of people, yet we have few treatments that are both safe and effective,” said Robert Modlin of UCLA, a co-author of the paper. “Harnessing a virus that naturally preys on the bacteria that cause pimples could offer a promising new tool.”

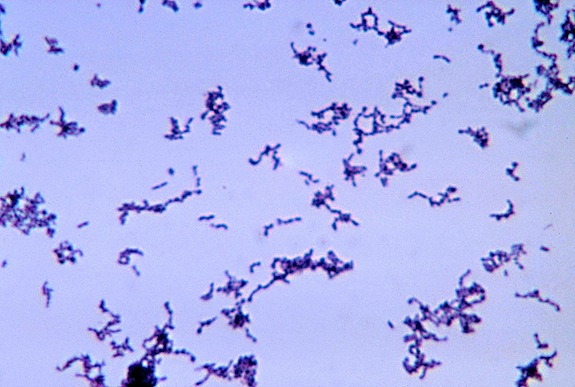
An electron microscope magnification of P. Acnes phages, the virus that naturally infects and kills P. acnes bacteria. Image via the University of Pittsburgh
Acne is caused by blockages in the skin’s follicles formed by an oil called sebum, which is produced by the body to prevent hair follicles from drying out. When sebum forms a plug in the follicle, it allows the naturally occuring bacteria P. acnes to trigger an inflammatory response in the skin, leading to swollen red bumps and other symptoms. Antibiotics and other prescription acne treatments work by killing these bacteria, but over time, antibiotic-resistant strains of the bacteria have emerged, rendering these products less effective.
The research team decided to explore the potential of an entirely different method—killing the bacteria by using a type of virus that also lives naturally on the human skin and has specifically evolved to infect P. acnes bacteria. To do so, they gathered both the bacteria and 11 different versions of the virus (P. acnes phages—named for the host bacteria it preys upon) from the faces of volunteers using over-the-counter pore-cleaning strips.
An analysis of the different viruses’ DNA, as collected from the volunteers, revealed surprisingly little genomic diversity (all samples were identical for at least 85 percent of their DNA)—a trait that would make developing an acne treatment simpler because it indicates that any formulation of the virus would be effective in killing the P. acnes bacteria for many different people. This conclusion was bolstered by the fact that, when the researchers cultured bacterial samples from different volunteers and added the varieties of virus, the viruses were effective in killing a broad range of different sub-varieties of bacteria.

The clear spots in this cultured colony of P. acnes bacteria indicate where the virus was effective in killing it. Image via UCLA/Modlin Lab
Additionally, the specificity of the virus’ killing mechanism makes it an appealing candidate for an anti-acne treatment, in contrast to antibiotic treatments that can also harm populations of beneficial varieties of bacteria that live on our bodies. “Phages are programmed to target and kill specific bacteria, so P. acnes phages will attack only P. acnes bacteria, but not others like E. coli,” said lead author Laura Marinelli of UCLA. “This trait suggests that they offer strong potential for targeted therapeutic use.”
The researchers believe that the key to the virus’ killing ability is an enzyme it produces called endolysin, which may act by breaking down bacterial cell walls. A better understanding of how this enzyme works is a next step towards eventually developing a treatment, either based on endolysin isolated from the virus, or using the virus itself.
The team plans to test endolysin on its own to determine if can kill P. acnes bacteria on its own, without the virus. If the enzyme is successful in petri dishes, they may proceed by testing an extract made from the virus on participants to see whether it is a safe and effective way to prevent acne in human skin.
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/accounts/headshot/joseph-stromberg-240.jpg)
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/accounts/headshot/joseph-stromberg-240.jpg)