The Physics of Whisky’s Aesthetically Pleasing Residue
A photographer teamed up with scientists to figure out the fluid dynamics behind patterns left in whisky glasses
Plenty of souls have searched for answers at the bottom of a glass of whisky. For Phoenix-based artist and photographer Ernie Button, that quest revealed some unexpected beauty, and set him out on a search for truth.
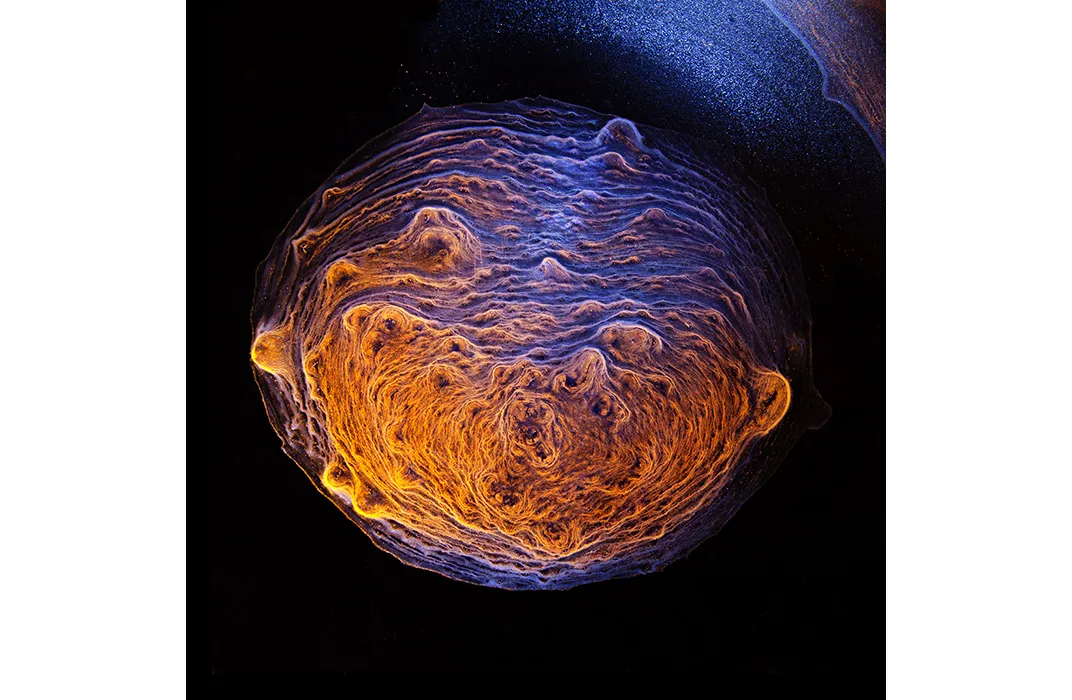
Over the last few years, Button has been capturing stunning images, like the ones seen above, of the dried patterns that whisky leaves at the bottom of a glass. Recently he teamed up with Howard Stone, an engineer at Princeton University, whose lab found that some basic fluid dynamics drive whisky’s unique pattern formation. They presented their findings today at a meeting of the American Physical Society (APS) in San Francisco, California.
Button’s fascination with whisky began when he married into his wife’s Scotch-drinking family. While doing the dishes at home, he noticed that lacy lines covered the bottom of a glass of single-malt scotch. Other glasses appeared to produce various patterns of dried sediment. “It’s a little like snowflakes, in that every time the Scotch dries, the glass yields different patterns and results,” says Button. He thought that trying to capture the patterns might make for an interesting photography project.
Creating the images required a bit of Macgyvering. On their own, the grayish sediment lines are a bit underwhelming compared to the amber liquid that creates them, so Button had to experiment with different glasses and lighting systems. Using flashlights and desk lamps, Button highlights the patterns with different hues. “It creates the illusion of landscape, terrestrial or extraterrestrial,” says Button. To him, many of the images appear celestial, perhaps something that a satellite camera might snap high above Earth. Other images could easily be frigid polar vistas or petri dishes of bacterial colonies.

Button captured a lot of variety through his camera lens, and he began wondering if it had something to do with the age of the liquid. After some experimenting, though, he saw little difference in younger and older versions of the same type of whisky. With some Googling, he came across Stone’s lab, then at Harvard and now at Princeton. Stone and his colleagues happily answered questions over email, and the conversation got them thinking as well.
Stone initially suspected that something called the coffee ring effect might be at play: When coffee dries, particles are pulled to the edge of where the liquid makes contact with the cup, creating ring-like patterns as the water evaporates. Similarly, the differing evaporation tendencies of alcohol and water can create interesting patterns, like the "legs" on a wine glass. This is largely driven by the Marangoni effect, first described by 19th-century physicist Carlo Marangoni. Alcohol and water have different surface tensions—that’s the degree of attraction liquid molecules have to other surfaces (in this case a cup or a glass). Alcohol has a lower surface tension than water, and alcohol evaporation drives the surface tension up and pushes more liquid away from areas of high alcohol concentration.
In the case of whisky, the patterns were more uniform, with particles settling in the middle of a droplet of liquid. So was there something about whisky that created unique patterns compared to other types of liquors?
Not a whisky drinker himself, Stone ran to the store to buy a bottle or two, and his team began tinkering around in the lab. Under the microscope, they made videos of whisky drying and compared them to videos of a mixture of alcohol and water that mimics the proportions of whisky (about 40 percent ethanol, 60 percent water). The fake whisky followed Marangoni flow: Ethanol evaporated first, drawing the particles into a ring-shaped pattern. The higher the alcohol content, the smaller the ring. But whisky, as Button had observed, didn’t produce clean rings. “That says there’s something in your mixture that’s missing,” explains Stone.
Next they added a soap-like compound, which sticks to the surface of water, to their faux-whiskey. Lots of compounds can do that, so they thought whisky might contain something similar. But the patterns still weren’t quite right. Next they added a larger molecule (a polymer) that might help whisky stick to the surface of the glass. Finally, the mixture droplets were doing roughly the same thing as whisky droplets.
Based on this work, the lab team has a hypothesis: “Very small amounts of the additives that come from how whisky is made contribute to the kinds of patterns you actually see,” says Stone. Different additives or variations in the manufacturing process might possibly produce different patterns.
The research has some practical implications. Better understanding of these types of fluid flows could prove useful in a lot of industrial situations that involve liquids, particularly liquids that contain particles of sediment or other material, such as printing inks. In the meantime, Button hopes his images raise questions in the minds of viewers that may give them some interesting conversation starters at cocktail parties. “The science behind the imagery provides that extra added layer of thought and complexity,” he says.
For more images and information about Ernie Button’s work and upcoming exhibitions, check out his website.
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/accounts/headshot/Screen_Shot_2014-01-27_at_12.05.16_PM.png)










/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/accounts/headshot/Screen_Shot_2014-01-27_at_12.05.16_PM.png)