Extinction or Evolution? The Answer Isn’t Always Clear
The same factors that kill off some species cause others to evolve at lightning speed
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/74/68/74689c14-604d-48ec-8dc1-49c1ac1595de/cnx1w9.jpg)
One fish, two fish, crayfish—new fish?
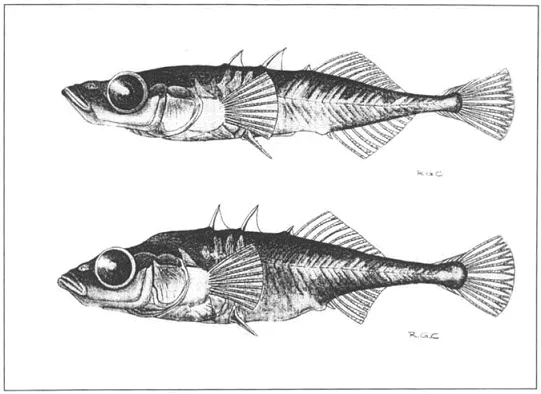
Though it might sound like the plotline of a Dr. Seuss book, that’s what actually happened to the threespine stickleback fishes of Canada’s Enos Lake. For thousands of years, two distinct species of these spiny silver sea creatures—known as the benthic sticklebacks and the limnetic sticklebacks, both descended from a single species—lived in peaceful coexistence. The former stayed near the floor, where they fed on bottom-dwellers; the latter swam up near the sun, eating insects at the surface. Their habitats and behaviors were so different that they rarely met, and never interbred. And all was well.
But then something strange happened: The two species of fish once again became one. How?
The answer had to do with invasive crayfish, which were likely introduced into the lake ecosystem by humans. Within three years of the crayfishes’ arrival, the two species had once again merged. “It seems like someone may have introduced the crayfish possibly as a food source,” says Seth Rudman, an evolutionary biologist at the University of British Columbia whose paper on the phenomenon came out in Current Biology earlier this year. “The crayfish physically altered the way the sticklebacks nest and breed, which increased the probability of mating” between the two species, he says.
You might be saying to yourself: Wait, that’s not how evolution works. Actually, it can be. What happened with the finger-length fishes is an example of “introgressive extinction,” otherwise known as reverse speciation. Regular speciation happens when members of one species are divided by changes in their habitat or behavior. The most well-known example is Darwin’s finches: Over time, finches on different, isolated islands diverged in beak size and other qualities until they became distinct species. Reverse speciation is when those distinct species come together again, until they become one species yet again.
But the process isn’t as simple as it sounds. Just because two become one, doesn’t mean it’s the same species that you started out with. With the sticklebacks, it still isn’t clear whether the new species is the same as the common ancestor that both evolved from, or something wholly different. “Whether they went back to being an ancestral species is debatable,” Rudman says. “Their ecological role is slightly shifted from the earlier species. It’s really hard to say if they’re going all the way back, but it might be following that path.”
Though it may sound counterintuitive, evolution and extinction aren’t mutually exclusive. Quite the contrary: Extinction was a fundamental part of the theory that Charles Darwin, the grandfather of evolution, put forth in 1861 in his seminal Origin of Species. “So profound is our ignorance, and so high our presumption, that we marvel when we hear of the extinction of an organic being; and as we do not see the cause we invent cataclysms to destroy the world, or invent laws on the duration of the forms of life!” he wrote. It’s obvious truth even if it seems contradictory: the same selective pressures that drive some species to extinction force others to adapt and evolve.
Today some researchers say that reverse speciation may be becoming more common—especially in environments altered by humans. Examples of this kind of hybridization abound: The endemic grey ducks of New Zealand are threatened with extinction not only from habitat loss, but also because of interbreeding with invasive mallard duck species. The mallards now account for more than 80 percent of New Zealand’s dabbling duck population, and as they continue to breed with grey ducks, the latter comes ever closer to extinction. Then there are Darwin’s famous Galapagos finches. Three distinct species of finch on the island of Floreana were observed in 1947, but since then researchers have found only two species, and the genetic evidence points to reverse speciation.
As with the case of the sticklebacks of Enos Lake, these examples show human activity is driving hybridization around the world—whether we’re aware of it or not.
Why some survive while others don’t is a crucial question in the age of the Anthropocene, the Earth’s sixth great extinction. And it’s one we are far from answering. In the past 15 years, we’ve lost numerous species, including the Hawaiian Po’ouli (or Black-faced Honeycreeper), the West African black rhinoceros, Spix’s Macaw and the Madeiran Large White Butterfly. And those are just some of the few we know about. It’s impossible to measure the full number since scientists haven’t identified all species on Earth, but we’re estimated to lose 1,000 to 10,000 times the normal number of species to extinction.
But it’s possible that within that flood of extinctions, there might be a number of species learning to rapidly adapt and survive.
Consider a 2015 study that followed robot “species” in a computer simulation of evolution. After introducing multiple random mutations over the course of generations, the researchers simulated a mass extinction event by killing off 90 percent of the species. In the wake of the mass culling, the remaining species evolved far more rapidly and efficiently. Another study looked at a newly discovered ichthyosaur species (the group comprises marine reptiles who lived around the time of dinosaurs). The researchers saw a similar burst of species radiation after the Permian extinction, including a diverse assortment of ichthyosaurs. Evolutionary biologist Chris D. Thomas has even found that plant speciation is accelerating during the Anthropocene.
These studies point to the idea that human activity, which is driving this mass extinction, might also be driving rapid evolution. That’s not to say mass extinctions are good, of course—just that there might be a thin silver lining to them. “We know very little about this on short time scales,” Rudman says. “We don’t have a great grasp on how much gene flow will facilitate adaptation and how much will remove adaptation.”
By better understanding this process, we might also be able to prevent future extinctions.
“Imagine a campfire,” Rudman says. “A little bit of gene flow—or hybridization—is like adding a few sticks or one small log. It fuels adaptation. But if you dump a whole load of wood, it’ll just smother the fire out.” In other words, some hybridization might be helpful to prevent extinction—like in the case of the Florida panther. The big cats were on the verge of extinction, so scientists introduced cougars from Texas to breed with the panthers. Now the panther population is stable and genetically healthy again. But with the threespine sticklebacks there was too much hybridization, since both of the original species are now extinct. The separate roles they played in the lake ecosystem are no longer being filled, which is changing the lake.
Of course, if humans have learned anything from tinkering with the natural environment, it’s that doing so can often have adverse effects. But Rudman’s work on the rapid evolution of some species gives him hope for the planet’s future. “I would start by saying I’m concerned, and it’s already been documented that there’s tremendous extinction going on,” Rudman says. “Having said that, I would say that perhaps I’m more optimistic than your average biologist because I study how fast some species can evolve.” And that evolution, he says, might just save some from extinction.