In a patchy ten-acre tract of grass in Coronado, a hilly exurb northeast of the Costa Rican capital of San José, a weedy horse paddock and corrugated metal stable stand adjacent to a building of pristine laboratories and climate-controlled habitats. Through one door is a necropolis of dead snakes preserved in glass jars arranged helter-skelter on a counter, reminiscent of a macabre Victorian cabinet of curiosities. Through another is a sterile-looking white room full of humming scientific instruments.

The Instituto Clodomiro Picado, or ICP, named after the father of Costa Rican herpetology, is one of the world’s leading manufacturers of snake antivenoms, and the only one in Central America. The need for antivenoms is far more urgent than a person living in a developed nation blessed with a temperate climate might suppose. Globally, venomous snakebites kill roughly 100,000 people each year, mostly in South Asia, Southeast Asia and sub-Saharan Africa. In these regions’ poorer corners, local capacities for antivenom production are limited or nonexistent; the ICP has stepped in to help fill the gaps. Beyond meeting its own country’s needs, the institute has supplied or developed lifesaving antivenoms for victims on four continents, each treatment customized to protect against species that still pose lethal threats, from the West African carpet viper to the Papuan taipan.
At one time, snakebite deaths were common in Costa Rica, as Picado himself documented in his 1931 book Venomous Snakes of Costa Rica. He reported 13 in just one month—a death rate, given the population of about 500,000, higher than the current global death rate from lung cancer. Largely because of the ICP’s antivenoms, snakebite deaths in Costa Rica today are negligible, typically one or two per year in a current population of some five million—about the same per capita death rate as powered lawn mower accidents in the United States.
Celebrated for its abundance of tropical wildlife, Costa Rica is a place where it pays to watch your step. It is home to 23 species of venomous snakes, including the Central American bushmaster—one of the world’s largest vipers, growing up to 11 feet—and the bocaracá, whose indigenous name means “devil that brings death when it bites.” Yet none is more feared than Bothrops asper—the terciopelo, also known as the fer-de-lance. Across a range extending from Mexico to northern Peru, the terciopelo is dreaded for its tenaciously defensive temperament: In situations that would cause other vipers to flee, it strikes. And when the terciopelo bites, it injects a remarkable volume of venom, around ten times as much as a copperhead.

For the stricken, the result is hellish. Terciopelo venom destroys the flesh at the injection site, causing severe swelling, tissue death and excruciating pain. As it travels through the body, it induces internal bleeding and, in severe cases, organ failure and death. Blood can seep out of the nose and mouth, among other orifices, which Mayans compared to sweating blood. Picado described the late stages of such a snakebite this way: “If we ask the wretch something, he may still see us with misted eyes, but we get no answer, and perhaps a last sweat of red pearls or a mouthful of blackened blood warns us of the triumph of death.”
* * *
“Are you scared?” asked the ICP snake handler Greivin Corrales, with a touch of concern and some mild amusement. I was standing in a small room with a six-foot-long terciopelo, unrestrained on the floor, only a few feet away from me. Corrales had witnessed me tense up when he removed the snake from a bucket with a hook; I had heard of the terciopelo’s reputation. Corrales’ colleague Danilo Chacón referred to the specimen as a bicho grande, using an untranslatable term that falls somewhere between critter and beast. The snake exhibited the characteristic scale pattern of diamond and triangles in light and dark brown, and the trilateral head that inspires the common name fer-de-lance, or lancehead. Though the snake was highly conspicuous on the terrazzo tiles, the markings would blend seamlessly with Costa Rica’s forest floor, making it all too easy to step on such a bicho.
The ICP has mastered the process of antivenom production, and I had come to watch the fundamental first step: the extraction of venom from a live snake, sometimes called “milking.”
The bucket from which the snake had been drawn was full of carbon dioxide gas, which temporarily sedates the snake, making the process less stressful for both animal and handler. Chacón, the more experienced handler, only recently started using carbon dioxide after nearly 30 years working with unsedated terciopelos. “I think it’s about not getting overconfident,” said Corrales. “Once you’re too confident, you’re screwed.” Even while occasionally handling unsedated snakes, the technicians use bare hands. “You have to feel the movement,” he said. “With gloves you don’t feel the animal, you don’t have control.”
The handlers bent down and picked up the groggy terciopelo, Chacón grabbing the head, Corrales lifting the tail and midsection. They led the snake headfirst to a mechanism topped by a funnel covered with a layer of thin, penetrable film, which the snake instinctively bit. Venom dripped from the fangs, through the funnel and into a cup. In its pure form, viper venom is viscous and golden, resembling a light honey.

Antivenoms were first developed at the end of the 19th century by the French physician and immunologist Albert Calmette. An associate of Louis Pasteur, Calmette was stationed in Saigon to produce and distribute smallpox and rabies vaccines to local people. Alarmed by a surge of fatal cobra bites in the area, Calmette—who later gained fame as an inventor of the tuberculosis vaccine—applied the principles of immunization and vaccination to snake venom. He injected serial doses into small mammals in order to force their bodies to recognize and gradually develop antibodies as an immune response to the toxins in the venom. In 1895, he began producing the first antivenoms by inoculating horses with Asian cobra venom, drawing the horses’ blood, separating the venom-resistant antibodies, and mixing them into a fluid that could be injected into a snakebite victim.

Today, the ICP produces antivenoms in much the same way, but with more advanced processes allowing for a purer product. “Our antivenoms are basically solutions of horse antibodies specific against particular venoms,” said José María Gutiérrez, a former director of the ICP and a professor emeritus at the University of Costa Rica, which oversees the institute. The ICP’s roughly 110 horses live mostly on a farm in the nearby cloud forest and are brought to the stables to take part in antivenom production periodically. Venom is injected into a horse’s body in tiny amounts every ten days for two or three months initially, then once every two months—enough for its immune system to learn to recognize and create antibody defenses against the venom over time, but not enough to harm the horse. Afterward, blood is extracted from the horse in a quantity that is “like donating blood at a blood bank,” according to Gutiérrez. “We have the horses under strict veterinary control.”
Once the blood settles, the antibody-containing plasma is separated, purified, filtered, sterilized and mixed into a neutral liquid. The antivenoms are sent to hospitals, clinics and primary health posts, where they are diluted with saline and administered intravenously into snakebite victims.

Antivenom counteracts venom precisely on a molecular level, like a lock and key. Because venoms vary chemically among species, an antivenom to protect against a specific snake’s bite must be prepared with venom from that snake, or from one that has very similar venom. To produce an antivenom that protects against multiple species, called a “polyvalent,” different venoms must be combined strategically in production. “That specificity makes anti-venoms sort of difficult to produce,” said Gutiérrez. “In contrast, tetanus antitoxin is the same all over the world, because tetanus toxin is a single toxin.”
The ICP maintains a diverse collection of live snakes, mostly caught and donated by Costa Rican farmers and landowners, some bred in captivity. From these, the ICP technicians have built an impressive stock of extracted venoms, supplemented with occasional imports of exotic venoms.
“Venom, more venom, and more venom there,” said serpentarium coordinator Aarón Gómez, opening a freezer in a laboratory room, exposing dozens of samples. After extraction, most of the venoms are immediately dehydrated for preservation. He unscrewed the top of a plastic container the size of a spice jar, revealing contents that looked like yellow ground mustard powder. “That’s terciopelo venom,” he said. “We have 1.5 kilos,” he said with raised eyebrows. That’s enough to kill 24 million mice or probably thousands of people.
Where the Serpents Are
The snakes that produce the world’s most potent venoms inhabit deserts, tropical forests and warm seas. Many pose a grave threat to people, but others are seldom encountered. Below the map, learn about ten of the most lethal snakes, ranked in descending order by venom potency. —Research by Katherine R. Williams
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/08/e7/08e70f12-25ba-4893-a8d4-2154276646aa/nov20snakemapweb.png)
Enhydrina schistosa
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/57/0e/570e9c30-bd33-443d-90e2-f08e7070de75/1.jpg)
Lethal venom dose*: 0.6 micrograms
Venom yield**: 79 milligrams
Common name(s): Beaked sea snake, hook-nosed sea snake, Valakadyn sea snake
This highly aggressive species kills more humans than any other sea snake. Its venom is so potent that one animal may carry enough to kill as many as 22 people.
Dabola russelli
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/58/f1/58f167b4-d470-4f7f-9702-cb34f260b992/2-.jpg)
Lethal venom dose*: 0.9 micrograms
Venom yield**: 250 milligrams
Common name(s): Russell’s viper
Though an antivenom exists, thousands of people in Asia and India die each year, of cerebral hemorrhage or kidney failure, after encountering this snake in open country and farmland.
Oxyuranus microlepidotus
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/05/c6/05c693d1-270e-4911-be8c-e623c64a6a0e/3-.jpg)
Lethal venom dose*: 0.9 micrograms
Venom yield**: 11o milligrams
Common name(s): Inland taipan
An enzyme boosts venom potency by breaking down connective tissue, helping toxins spread throughout the body. Feeding primarily on rodents, it’s shy around people; no human deaths are recorded.
Aipysurus duboisii
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/04/53/0453f359-2a8f-49c3-ac07-5078f199d521/4-.jpg)
Lethal venom dose*: 1.2 micrograms
Venom yield**: 0.7 milligrams
Common name(s): Dubois’ sea snake, reef shallows sea snake
The second most venomous sea snake in the world produces a neurotoxin that causes muscular paralysis. Still, it has short fangs and a low venom yield.
Pseudonaja textilis
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/2b/58/2b58ff01-eac4-4ab9-a131-70ea13aa6992/5-.jpg)
Lethal venom dose*: 1.5 micrograms
Venom yield**: 67 milligrams
Common name(s): Eastern brown snake, common brown snake
Noted for speed and aggression, it nonetheless discharges a significant venom dose in just 20 percent of human attacks. One to two human deaths occur in Australia annually.
Dendroaspis polylepis
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/9e/0b/9e0b269a-007b-4643-a9e3-280bdf44c63d/6-.jpg)
Lethal venom dose*: 1.5 micrograms
Venom yield**: 100 milligrams
Common name(s): Black mamba
This snake, which lives in trees and on the ground in sub-Saharan Africa, can grow nine feet or longer. Its bite is usually fatal unless a victim receives antivenom. Death can occur within 20 minutes.
Crotalus tigris
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/55/a0/55a0dcf3-1130-4e3c-a243-930024991ad8/7-.jpg)
Lethal venom dose*: 1.8 micrograms
Venom yield**: 11 milligrams
Common name(s): Tiger rattlesnake
While it has one of the Western Hemisphere’s most lethal snake venoms, a low yield and fangs extending just a few millimeters combine to prevent severe attacks on humans.
Dispholidus typus
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/36/0a/360a6a30-02c8-422e-b79e-a4b1ca02e099/8-.jpg)
Lethal venom dose*: 2.1 micrograms
Venom yield**: 8 milligrams
Common name(s): Boomslang
Considered nonaggressive, this species of tree snake has fangs in the back of its mouth and is fully capable of killing people, with a venom that causes internal bleeding and kidney failure.
Hydrophis platurus
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/1c/35/1c351cf5-4b78-4482-83ac-2afc6e41354f/9-.jpg)
Lethal venom dose*: 2.1 micrograms
Venom yield**: 4 milligrams
Common name(s): Yellow-bellied sea snake, pelagic sea snake, black and yellow sea snake
The world’s most wide-ranging sea snake has a highly toxic venom that is delivered in small doses through short fangs. Human interactions are uncommon.
Bungarus caeruleus
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/fa/f4/faf452fb-159f-41d4-91c6-353ed247cda9/10-.jpg)
Lethal venom dose*: 2.7 micrograms
Venom yield**: 20 milligrams
Common name(s): Common Indian krait, blue krait
Believed to cause thousands of human deaths in Asia, the nocturnal reptile may seek shelter in houses in the rainy season. An untreated victim may become paralyzed and die of respiratory failure.
*Estimated amount of venom, in micrograms, to kill 50 percent of laboratory mice in a sample, if each mouse weighed 30 grams. A microgram is 0.001 milligram, roughly the mass of a single particle of baking powder.
**Maximum amount of venom, dried, in milligrams, produced at one time by an adult snake.
The ICP’s success in maintaining and breeding snakes that otherwise fare poorly in captivity has allowed for the collection to include workable quantities of exceedingly rare venoms. For example, an innovative technique involving a diet of tilapia filets sustains about 80 coral snakes in the serpentarium, a rare quantity. “Most other producers don’t produce coral antivenom,” said Gómez. “But because we have the snakes, we can produce the venom, so we can produce the antivenom.” A potent neurotoxin, coral snake venom is about four times as lethal as terciopelo venom. In powdered form, it is pure white.
* * *
There’s no question that historical factors like accessible health care, the migration from rural to urban areas, and even a decrease in barefootedness contributed to the decline of snakebite deaths in Costa Rica. But without the ICP’s antivenoms, bites would still carry a grave risk. Traditional remedies popular before the proliferation of antivenoms—such as drinking an elixir of tobacco leaf or rubbing a bone on the bite—were no match for snake venom.
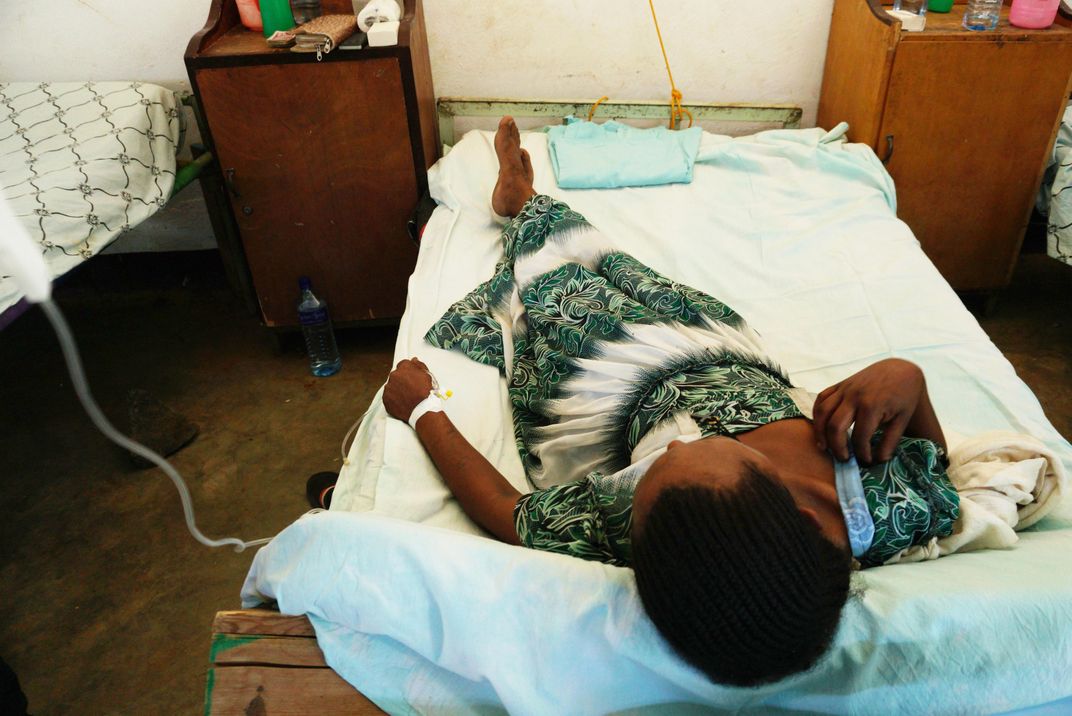
Other countries, however, cannot claim such progress. India alone suffers nearly 50,000 venomous snakebite fatalities each year, chiefly from the saw-scaled viper, the Indian cobra, Russell’s viper and the common krait. Nigeria’s snakebite mortality rate has been reported at 60 deaths per 100,000 people—more than five times the mortality rate from automobile accidents in the United States.

“We want to expand the knowledge and expertise generated in Costa Rica to contribute to solving this problem in other regions and countries,” said Gutiérrez, who is also a member of the board of directors of the Global Snakebite Initiative, a nonprofit that advocates for greater recognition and understanding of snakebite mortality worldwide, especially in impoverished regions. Since the near-eradication of snakebite deaths in Costa Rica, the ICP has endeavored to fill antivenom vacuums in these faraway places where antivenoms have been inadequate, inaccessible or nonexistent.
Even the United States, with its advanced medical science and robust pharmaceutical industry, has experienced occasional antivenom shortages. Despite the exorbitant prices for which the product can be sold in the U.S.—generally over 100 times what ICP antivenoms go for—the relative rarity of venomous bites and the esoteric, labor-intensive manufacturing process have kept antivenom production a niche industry there. Only two entities in the United States currently produce snake antivenoms for human use: Pfizer (to counteract coral snake venom) and Boston Scientific (to counteract pit vipers like rattlesnakes).

That leaves labs like the ICP fulfilling the supply of antivenoms where the demand is greatest. Founded in 1970, ICP began steadily furnishing the drugs to other Central American countries in the 1990s. To develop new antivenoms for regions in need, in the early 2000s it began importing foreign venoms with which to inoculate its own horses; the institute doesn’t import live snakes because of ecological and safety concerns.
For a decade the institute has been distributing a newly developed antivenom to Nigeria, capable of protecting against the venoms of the West African carpet viper, the puff adder and the black-necked spitting cobra. Bites from these deadly snakes had been treated in the past mostly with a polyvalent antivenom manufactured by Sanofi-Pasteur, but the French pharmaceutical giant, citing a lack of profit, ceased production in 2014, leaving a dangerous gap in the market. The ICP’s antivenom is now being used in other countries in the region, from Burkina Faso to the Central African Republic. “Doctors Without Borders is now using our antivenom at their stations in Africa,” said Gutiérrez.

“The Instituto Clodomiro Picado has been doing this production for many, many years, and they’ve got it dialed in,” said Steve Mackessy, a biochemist from the University of Northern Colorado, who has collaborated with the institute. “They produce an affordable product that works very, very well. So applying that to a situation where you have anti-venoms that either weren’t available at all, or were poor quality, or poor efficacy because they’re mostly designed against other species, that’s a godsend for those countries.”
An estimated 250,000 people have been treated with ICP’s antivenoms in Central America, South America, Africa and the Caribbean. The institute has recently developed new products for Asia, specifically Papua New Guinea—home to the extremely venomous taipan—and Sri Lanka, where imported Indian antivenoms used there have been described as largely ineffective.

Antivenoms may not be a lucrative business, but Gutiérrez stresses that access to such essential medicines should be considered a human right rather than a commodity. “This is a philosophical issue here,” he said. “Any human being that suffers snakebite envenomation should have the right to receive an antivenom.”
* * *
Clodomiro Picado himself—whose imposing bust adorns a sign outside the ICP’s entrance—was not generous in his estimation of the character of snakes. “He who dies victim of snakes does not fight, his death won not by conquest but by thievery,” he wrote. “For this reason the serpent, together with poison and the dagger, are signs of treachery and treason.” Gutiérrez is more measured, pointing out that snakes have been both gods and demons in mythologies around the world: “They’re fascinating, yet they can kill you.”
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/91/0b/910bc066-0381-4f2d-8f47-e0383d3d9b6c/mobile.jpg)
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/3a/17/3a177830-734c-44af-975c-f733ae24426b/1opener.jpg)