Let’s Build Cars Out of Batteries
If batteries could make up the very structure of our vehicles and electronics, those products would be far lighter and more efficient
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/79/e5/79e55e22-4d95-484f-8a5c-47d2b844ffde/car-with_titles.jpg)
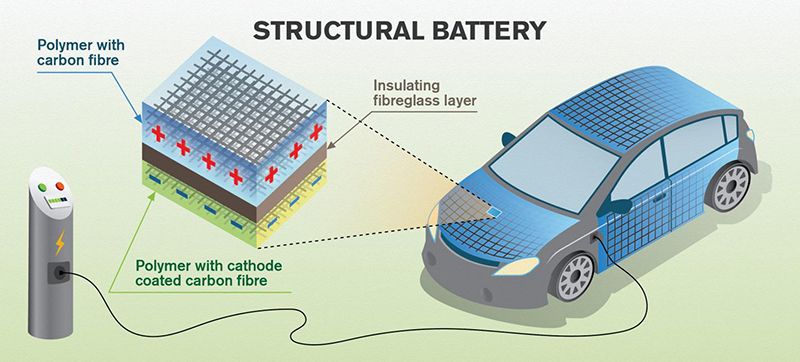
Carbon fiber is primarily used for its light weight, and prized for its strength and stiffness. But when Leif Asp looks at the material, he sees an opportunity to make it do double duty in a way that could drastically improve the efficiency of cars and airplanes.
“The battery is a structural parasite,” says the engineer and professor at the Chalmers University of Technology in Sweden, meaning it adds weight and saps efficiency, without contributing to the physical strength and structure of the car that it’s powering. But what if the vehicles were built out of batteries?
That’s where Asp is really going with this technology. He wants to see cars, planes, boats, even smartwatches and other consumer electronics made of a material that acts both as the body and the energy source—something known as a “structural battery.” A car incorporating structural batteries could weigh up to 50 percent less than a typical EV that has heavy lithium ion batteries packed underneath it, says Asp.
It’s not news that carbon fiber has electrochemical properties. Like graphite, the material is, in certain configurations, capable of conductivity. Researchers from the Chalmers University of Technology have applied for a U.S. patent on a battery made from carbon fiber, but actually bringing one to market has proven tricky for the small number of people studying the idea. New research from Asp’s team has identified a particular aspect of the material that makes its potential use as structural batteries a lot more realistic.

All carbon is not created equal, however, and different types of carbon have different properties that make them applicable to different uses. Asp’s goal is to understand what behaves how, and why, and apply that to structural applications.
“The carbon fibers that are available on the market, they have been made for structural applications or made for electrical applications,” he says. Structural applications are what we’re most familiar with, from the carbon that makes up bicycles and other strong, lightweight products, but electrical components are sometimes made of the material too, albeit a different type. He believes there’s carbon that can do both.
In their latest research, Asp and his collaborators compared three composites and examined them through electron microscopy and laser spectroscopy. They built the fiber into batteries, looked at the size and orientation of the crystals of carbon atoms bonded together in them, and compared the stiffness, strength and electrochemical properties of the different materials. Smaller crystals, with more disoriented structure, tend to be more electrochemically reactive — that is, they are more able to take up, store and release electrons, and thus act as batteries. However, these types of carbon are less stiff than those with crystals that are longer and lined up. (Either way, they’re very small; Asp compared fiber with crystals from 18 to 28 angstroms to crystals from 100 to 300 angstroms, and an angstrom is one ten billionth of a meter.)
Using a carbon fiber that sacrifices some stiffness to achieve better conductivity may not be a problem, because the material was still stiffer than steel, and capable of carrying a structural load. It also won’t hold a charge as efficiently as traditional batteries, but then, if most of the car is made up of the stuff, it won’t have to because the overall efficiency will still be greatly increased. Industry partners like Airbus, which has been working with Asp since 2015, refer to this as “mass-less energy storage.”
Still, it’s technology that’s a long way from being practical — potentially decades, says Adrian Mouritz, executive dean of the school of engineering at RMIT University in Melbourne. Mouritz also works on structural energy storage using carbon fiber, but his work embeds lithium ion batteries within sandwiches of carbon, helping to carry some of the structural load and reducing the dead weight of batteries, though not as extensively as Asp’s version.
“The approach we’re taking, the composite material is already proven, the battery itself is already proven. All we’re trying to prove is the integration of the battery into the composite, which is a much smaller step to take,” says Mouritz. “Leif’s is … more technically complex, but its benefits in the longer term are going to be stronger. It still just requires a lot more research and development to optimize the materials and design of the actual system.”
Asp and his lab are working to make it viable already. Early research (2014 and prior) modified carbon fibers, introducing a sheath of laminated polymer electrolytes that helps the fiber store and release ions more efficiently, in the same way that a lithium ion battery uses an intervening electrolyte.
“For this to fly, of course, would be a long time away,” says Asp. He is working with Airbus to produce a demo, for release next year, which replaces interior lights and cables with structural carbon fiber. Though the greater weight savings could be in eliminating the need for fuel, which Mouritz says accounts for one third or more of an airline’s operating budget, the Airbus demo will be an illustration that the technology is viable.
Mouritz sees the technology being applied to luxury autos and Formula 1 racecars first, and wide adoption in the consumer market once the price comes down and the reliability is confirmed. “If you can lightweight your aircraft, if you can lightweight your automobile, the actual net cost savings of this is in the hundreds of millions if not in the billions of dollars,” he says.
“The other thing, of course,” Mourtiz adds, “is, if I’m reducing my fuel burn, I’m actually reducing my greenhouse gas emissions.”