Researchers Transfer a Human Protein Into Plants to Supersize Them
While a promising route to boosting crop yields, experts say more work needs to be done to understand why the tweak works
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/a0/c5/a0c534a5-f364-455c-ac4a-f701ab56dbdb/rice_plant.jpg)
Every year, 9 million people in the world succumb to hunger—that’s more than the deaths from AIDS, malaria and tuberculosis combined. But the solution to the global hunger crisis isn’t as simple as ramping up agricultural production.
For one, agriculture comes with a hefty carbon footprint, contributing 10 to 20 percent of the global greenhouse gases annually. Then, chemical overuse sows resistance in the pests and pathogens the substances intend to kill, making it harder for farmers to eradicate their associated diseases the next time around. Runoff from fertilizers may also pollute waterways.
It’s not enough to grow more food—humanity needs to grow more efficiently.
With an eye toward feeding the world’s growing population in a sustainable way, researchers from China and the U.S. may have found a clever solution—in the form of supersizing crops. Transplanting a human protein, known for promoting growth, into crops may engender larger, heavier and more bountiful plants, boosting agricultural yields by a whopping 50 percent, according to the new study in Nature Biotechnology. While the results are promising, experts say that more research needs to be done to test their agricultural mettle and ensure that the gains are replicable.
“We think this is a very good strategy to engineer our crops,” says study author Guifang Jia, a chemical biologist at Peking University in Beijing, China. But she admits “this paper is an initial study.” She wants to run many more follow-up tests, including safety evaluations, before the team’s prodigious produce can end up on the dinner tables of everyday consumers.
The protein responsible for the plants’ extraordinary growth spurt is the human fat mass and obesity-associated protein called FTO. While its associated gene gets a bad rap for increasing one’s obesity risk, the researchers previously reported that the protein is important for regulating growth in humans and other mammals. According to the researchers, FTO chemically modifies RNA strands, which are the short genetic recipes for individual proteins copied right out of the DNA playbook. This modification forces the RNA to produce the protein it encodes for. Essentially, FTO acts as a master “on” switch that ramps up widespread protein production across multiple RNA strands.
Several authors of the study had spent a decade understanding the role of FTO in humans. The researchers wondered what would happen if this RNA-targeting, growth-promoting protein made its way into plants instead.
“It [was] really a bold and bizarre idea,” says study author Chuan He, a chemist at the University of Chicago. The human body produces tens of thousands of proteins, and the first one his group tried in plants was FTO. “To be honest, we were probably expecting some catastrophic effects.”
Plants do not have an FTO-equivalent protein, says He. The chemist suspects that plants usually keep growth under a tight rein to prevent any one molecule from single-handedly wreaking havoc on a plant’s physiology. But in the face of a foreign protein, He says that plants lack any checks and balances to temper the consequences, whatever they may be. To the researchers’ surprise, FTO doesn’t kill off or cripple the plant. Instead, it forces plants to do the exact opposite: size up.
“[FTO] comes in, and there's no restriction to where it can access,” says He. “It’s a bomb.”
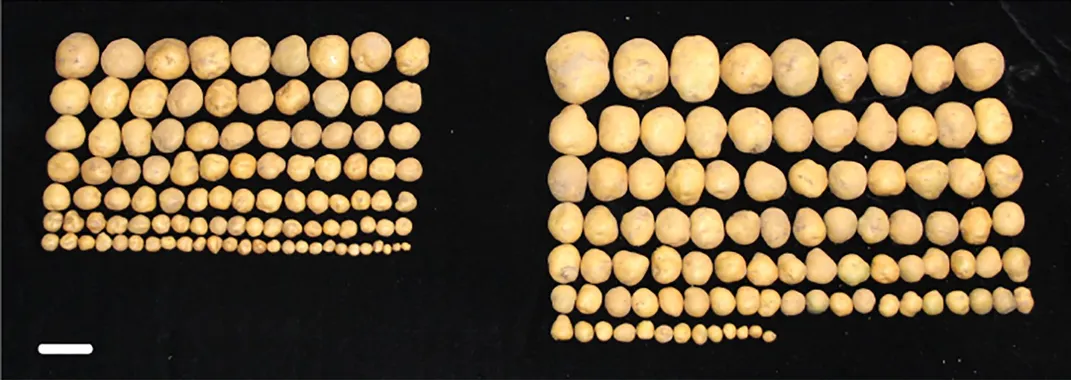
The researchers implanted the human FTO gene into the genomes of rice crops, allowing the plants’ protein machineries to take over and churn out this human molecule on its own. In the greenhouse, the genetically modified crops produced three times more rice than the untweaked plants. Grown out in fields in Beijing, the rice plants were 50 percent heavier and more productive. The same genetic modification also upsized the tubers of potato plants. Plants spiked with FTO also grew longer roots, showed higher photosynthesis levels and were more resistant to droughts.
Every type of plant the researchers altered, including grass and trees, grew faster and bigger. “This phenotype [trait] is consistently in any plant we engineer,” says Jia.

Despite its promise, genetic modification hasn’t been able to reliably amplify crops yields by more than 10 percent. Plant growth is complicated, genetically speaking—there simply isn’t one single gene to tweak. The study’s jaw-dropping percentage increases are amazing, almost unbelievably so, and other plant researchers recommend caution before getting any hopes up and ascribing more power to the protein.
“What's unusual about the strategy taken in this paper is that there is no reason to expect that it would have been successful,” says Donald Ort, a plant biologist at the University of Illinois at Urbana-Champaign who didn’t participate in the study. Science usually inches forward by building upon well-understood theories, says Ort. But the authors of the study seemed to have taken a stab in the dark and struck gold. “My guess is they were pretty surprised,” he says.
The study’s remarkable percentage increases in yield are much higher than the blips of several percentage points other researchers usually squeeze out, say Robert Sablowski, a plant biologist at the John Innes Center in the U.K. who wasn’t involved in the study.
“The final yield of a crop is genetically a very complex trait,” he says. Plants are constantly shifting priorities between bulking up or beefing up their defenses against diseases and stretches of unfavorable growth conditions. A plant’s surroundings usually have a large say in determining the final yield, but the study’s result seems to override most of these influences. “Because of all these compromises, it's very unusual to read a report of a very high yield increase with a simple genetic change,” Sablowski adds. “But it’s possible.” He says he would like to see the results replicated in different kinds of environmental conditions, and to better understand how the human FTO protein upends the carefully calibrated inner workings of a plant.
By absolute numbers alone, the FTO-modified rice and potato plants reported in the study have lower yields than commercial varieties, says Ort. Only if the same FTO trick increases the productivity of these already high performing strains, then that would be a true breakthrough. “The potential importance of this to agriculture still remains to be proved,” says Ort.
Both Ort and Sablowski are cautiously optimistic, because the path forward is clear: repeat the experiments elsewhere and with the best crop varieties humanity has to offer.
Jia and He are already on the chase—they’re working with farmers and probing deeper into the genetic mechanisms. But these experiments will take time, says Jia. Her team lost a growing season to last year’s lab shutdowns from the pandemic. She estimates her team will need several years to collect enough seeds from the handful of genetically tweaked parents to sow entire fields with their progeny.
The team is also experimenting with other non-plant proteins. So far, they’ve tried one other candidate, but didn’t see the same crop yield increases as wrought by the FTO protein. “I suspect it’s probably not going to work,” says He of other proteins. The regulatory pathways of plants are usually robust; it’s not common that scientists find a bug in their code that brings about biological bedlam. “I think FTO is very special,” says He.
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/accounts/headshot/kim.png)
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/accounts/headshot/kim.png)