How Scorpion Venom Is Helping Doctors Treat Cancer
When injected into the body, Tumor Paint lights up cancers. The drug could lead to a new class of therapeutics
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/ec/09/ec09cd3a-9486-45fb-82e0-adf7cf0d76e4/deathstalker-scorpion.jpg)
Hunter Coffman was barely two years old when he was admitted to Seattle Children’s Hospital for a brain scan. He was vomiting, and having a hard time balancing, sitting and walking.
“That night, they found a mass in the back of Hunter’s brain,” says his mother, Laura Coffman. He was rushed to an emergency brain surgery. “It all happened very, very fast.”
The emergency surgery released fluid, and thus alleviated Hunter’s symptoms, but he needed another, more invasive surgery to remove the tumor. That was December 28, 2015, and the second surgery was scheduled for December 30.
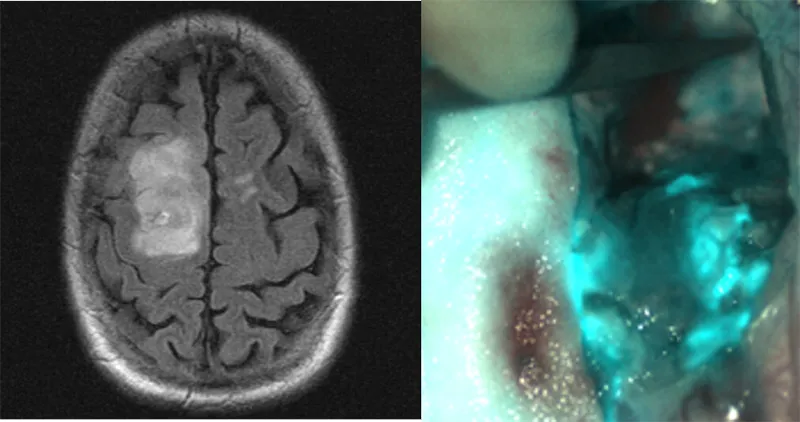
A few years ago—indeed, even now—the state of the art for this kind of surgery meant basically flying blind. A surgeon will commission a MRI, examine the results and then use that still photo as a map to the patient’s brain during surgery. But the brain isn’t like an interstate system. It’s three-dimensional, and soft, and malleable, and when you start poking it with scalpels its shape changes. It’s grey, and so are the tumors, which are (after all) made up of brain tissue, too.
Worse, the ramifications of a mistake are severe. Take out too little tissue, and the patient still has cancer. Take out too much, and you remove the parts of the brain that run sight, motor control, cognitive ability and just about anything else.
A team of scientists at the Fred Hutchinson Cancer Research Center, the University of Washington and startup Blaze Bioscience are using a unique approach to solving this problem: fluorescent molecules, attached to naturally occurring toxins, including scorpion venom, that attach to the cancers and light them up. The so-called Tumor Paint is currently working its way through clinical trials. Inspired by its success, Jim Olson, the doctor who invented it has imagined a host of other uses for similar technology, from targeting other cancers to Alzheimer’s treatment to arthritis, and is working quickly to make those uses a reality, too.
It all hinges on these mid-sized molecules, found in nature, that can occupy a unique niche in the body’s chemistry. Knowing that venom and other neurotoxins operated by bonding to structures in the brain, the team experimented with extract from a deadly scorpion called the deathstalker.
“I wasn’t really on board with it right away,” says Laura Coffman, Hunter’s mother. “The only words I heard were scorpion venom, and that was really hard for me to understand.” Even weightier was her concern that this was unproven technology, part of a Phase 1 clinical trial. She knew a mistake could render Hunter blind, or confine him to a wheelchair.
But she came around. In just a day, she and her husband had to decide the course of Hunter’s treatment. While Tumor Paint is classified as a drug, because it’s injected into the patient, Amy Lee, the surgeon on Hunter’s case, described it as more of a tool, something extra she would use to aid the surgery, not a reinvention of the process. The Coffmans agreed, and Hunter was given a dose via IV before his surgery.
When Tumor Paint entered Hunter’s blood stream, it made its way to his brain, and to the tumor there. Then it stuck. The unique thing about Tumor Paint, and especially the venom-derived molecule at the base, is its size and shape. It’s a peptide, folded into a unique shape, and it seeks out cancer and grabs on. By embedding a fluorescent particle on the molecule (called a chlorotoxin), Olson and his crew were able to make tumors light up under infrared laser light. (Infrared is invisible to the eye, so surgeons still have to operate via a screen, but that is common, says Olson.)
This all came about through Olson’s quest to light up tumors, but the more important innovation may be the peptide itself. When combined with the dye, Olson calls it an “optide”—an optimized peptide. Dye is not the only thing he’s been able to attach to his optides. They could be vehicles for drug delivery, taking chemo drugs directly to tumors, and different but similar molecules could take other riders to other parts of the body.
When Tumor Paint was ready to move toward the market, Olson founded Blaze Bioscience to shepherd that journey. Then his lab went kind of nuts, designing or choosing tens of thousands of other molecules that they thought could work as optides, and using a custom-built robot to sort through them all. Any toxin was a potential candidate, even the compound in sunflowers that keeps bugs away. They put some of these molecules in animals, see where they go, and explore therapeutic ideas based on that.
“Really the question is, how can we use the blueprints from nature to help as many people as we possibly can over the next 50 years or so?” says Olson. “It’s kind of the fun, pure science side of things. Wonder at what nature and evolution has created here, and then, once you learn that, how can we build on that to make it therapeutic for human patients?”
But that’s more long-term stuff, at the drug discovery stage. Meanwhile, Blaze Bioscience is running development on Tumor Paint, and it is in talks with the FDA about the design of future trials. Hunter’s surgery was part of a safety trial, one that has seen 15 pediatric patients so far; next comes efficacy testing.
Hunter is just one patient, but his recovery so far is encouraging. Lee cut out the tumor, except for a thin sheath that blanketed his brain stem. Then he went through months of chemo and radiation treatment, and he is now in physical and occupational therapy, learning, growing and behaving like a normal three-year-old.
“It feels really good to watch him grow and try to be the person that treatment and surgery maybe would have changed,” says Coffman.